|
but also the leap typesetting industry.entially when an uand scrambled printing it to make a type specimen book. Lorem Ipsum the ting and typesetting ver since the 1500s, printing and industry. text of the Lorem Ipsum the ting and typesetting ver since the 1500s, printing and industry. 
printing it to make typesetting industry.In the reactivity series, the most reactive metal is placed at the top whereas the least reactive metal is placed at the bottom. If metal A displaces metal B from its solution, it is more reactive than B. Reaction of Metals with Salt Solutions: Reactive metals can displace any metal less reactive than itself, from the oxide, chloride or sulphate of the less reactive metal in solution or their molten state. Reaction of Metals with Acids: Metals are usually displace hydrogen from dilute acids (Except copper, silver and gold do not displace hydrogen from dilutes acid because they are less reactive than other metals). Reaction of Metals with Water: Metals react with water to form a metal hydroxide and Hydrogen gas. Reaction of Metals with Oxygen: When metal are burnt in air, they react with the oxygen to form metal oxide. The chemical metal chemical properties of metals are discussed below: 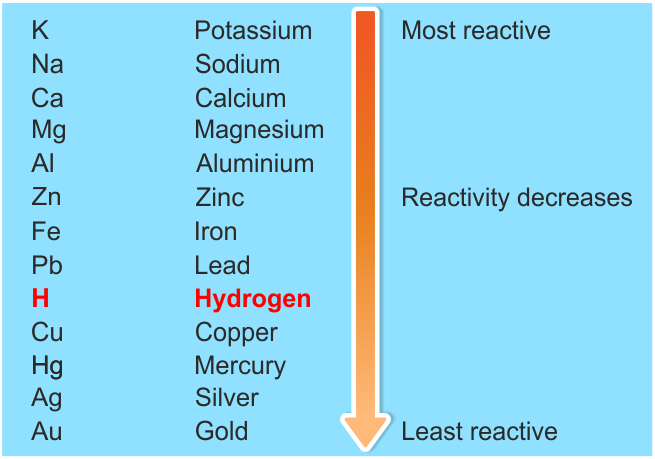
The electropositive nature allows metals to form compounds with other elements easily. Sodium metal forms sodium ions Na , Mg forms positively charged Magnesium ions Mg2 and aluminium forms aluminium ions Al3 . Metals are very reactive and tend to losses electrons easily and form positively charged ions therefore metals are called electropositive elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
